QIMA Life Sciences offers a range of canine in vitro and ex vivo models developed to support veterinary research and product evaluation. These 3Rs-aligned platforms include canine primary cell assays in both 2D and 3D formats, our fresh canine whole blood tests, and more. Such assays can be used to investigate a variety of pathological contexts and biological stressors, including: inflammation, infection, senescence, degeneration, atopic dermatitis, wound healing, and other stress-related responses.
Our ex vivo canine whole blood bioassays are designed for the rapid assessment of immune and inflammatory responses in a complex native matrix, with samples processed within hours of collection to support a robust turnaround time. By preserving interactions between circulating cell populations and soluble mediators, this model offers a valuable approach for studying compound activity, mechanism of action, systemic safety, and translational biomarkers in a physiologically relevant veterinary context.
The results presented below highlight findings from a case study using this canine whole blood model. Please do not hesitate to contact us for more information.
Objective of the canine whole blood study:
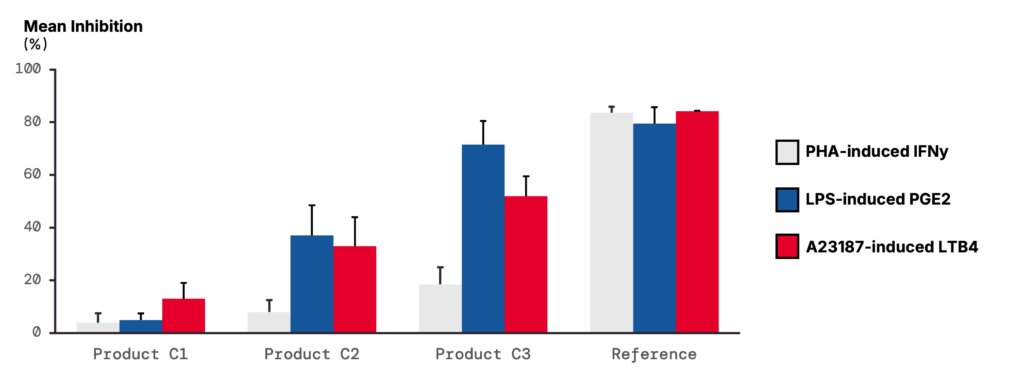
The aim of this study was to evaluate the anti-inflammatory and immunomodulatory effects of a product at three different concentrations (C1, C2, and C3). The production of IFN-γ was quantified as an indicator of T-lymphocyte activity, COX-2 activity was assessed by PGE2 measurement, and 5-LOX activity was measured through LTB4 levels. The study was performed on whole blood from five individuals.
1 – Complete blood count: haematological parameters measured prior to functional readouts.
Complete blood cell counts and related blood parameters were measured using the scil Vet ABC haematology analyser, programmed for canine parameters. This provided an initial assessment of the whole blood samples before functional testing.

2 – T-lymphocyte activity: PHA stimulation increases IFN-γ release, with only a slight effect of product pre-treatment.
T-lymphocyte activity was evaluated through IFN-γ quantification. In unstimulated conditions, IFN-γ remained below the limit of quantification; following PHA stimulation, release increased to > 1,000 pg/mL. Pre-treatment with the product produced a slight dose-dependent reduction in this induced response.
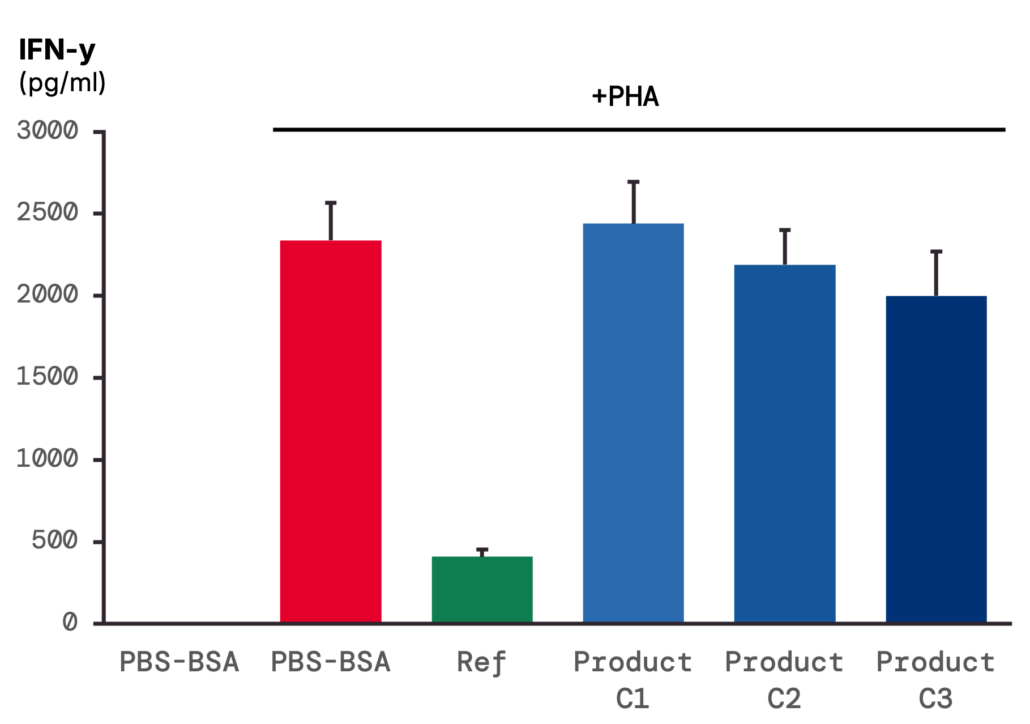 –
–
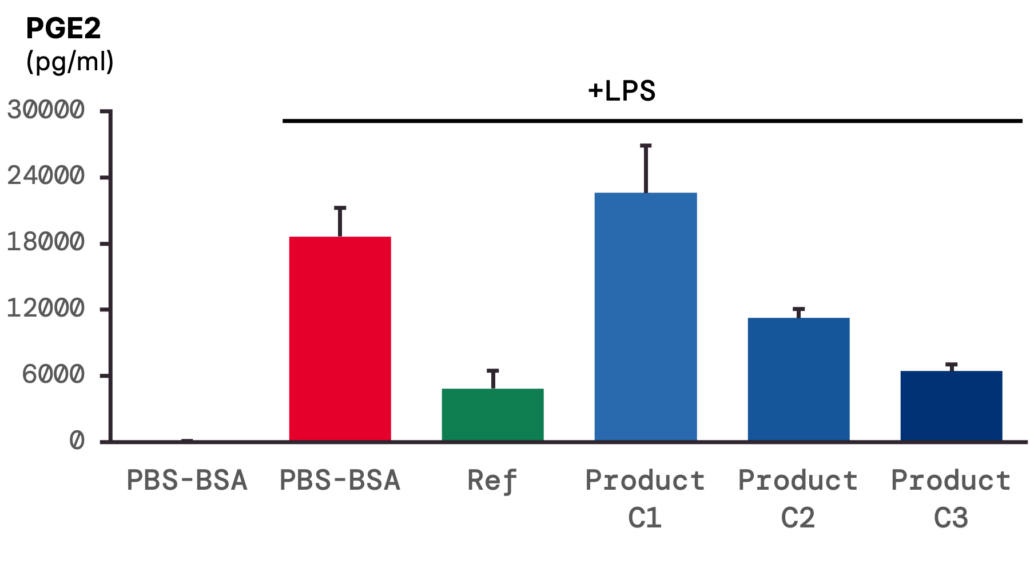
3 – COX-2 activity: LPS stimulation increases PGE2 release, inhibited by product pre-treatment in a dose-dependent manner.
COX-2 activity was assessed by measuring PGE2. Basal production was very low in the absence of stimulation, whereas LPS induced a marked increase, with levels exceeding 100,000 pg/mL. Product pre-treatment reduced the LPS-induced PGE2 production in a dose-dependent manner and, at the highest concentration (C3), was as effective as the reference treatment.
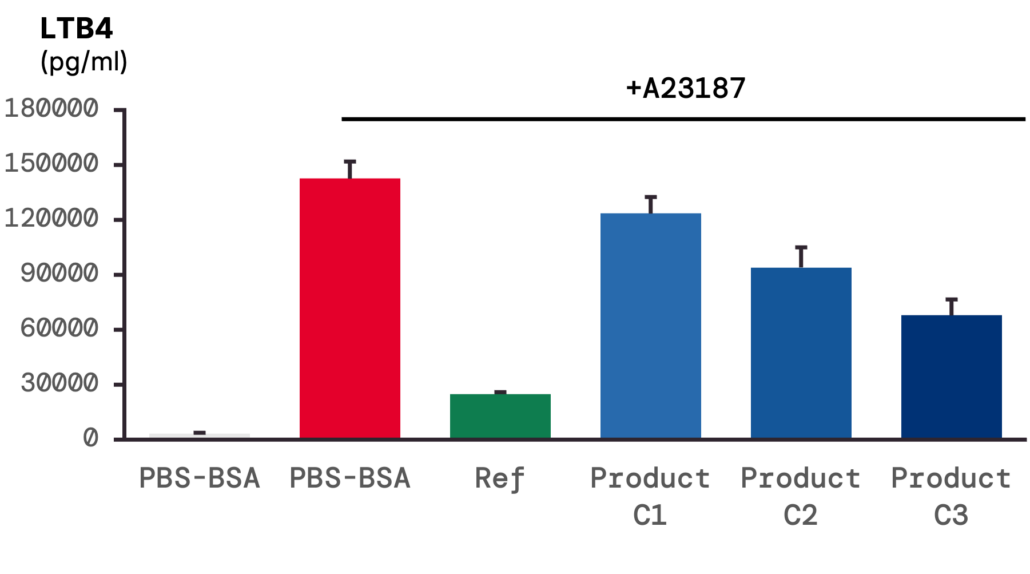
4 – 5-LOX activity: A23187 stimulation increases LTB4 release, inhibited by product pre-treatment in a dose-dependent manner.
5-LOX activity was evaluated through LTB4 measurement. In unstimulated samples, production remained low, but A23187 stimulation increased LTB4 release to > 90,000 pg/mL. Pre-treatment with the product inhibited this response in a dose-dependent manner.
See more on our Canine
Whole Blood Assays
Summary
The canine whole blood model provided robust functional readouts for T-lymphocyte responses, COX-2 signalling and 5-LOX activity. Blood cells remained responsive to stimulation, and the reference compounds inhibited more than 80% of the response for all dogs across the conditions tested.
In this study, the product reduced PGE2 and LTB4 release in a dose-dependent manner, while having only a slight effect on PHA-induced IFN-γ release, suggesting a degree of specificity in its mode of action. These data demonstrate one of several uses of the canine whole blood model, in delineating anti-inflammatory and immunomodulatory treatment effects in a low-manipulation system.