See more on our Canine
Whole Blood Assays
in silico virtual screening
QIMA Life Sciences offers an in silico screening tool (SELNERGY) to identify potentially active molecules for a given biological target or conversely. This tool makes it possible to screen several molecules or biological targets of interest in a computerized way, to classify and select the best candidates in order to rationalize the in vitro assays to be performed later.
in silico virtual screening makes it possible to predict the biological activity of a molecule of interest and define its fields of application, as well as to find the best compounds able to modulate the biological activity of a target. Learn more about in silico virtual screening.
Canine 2D in vitro models – Primary cells
Our cell bank has a panel of primary cells that target the skin and the osteoarticular system:
- Canine keratinocytes
- Canine fibroblasts
- Canine chondrocytes
- Canine osteoblasts
These cell models were isolated from samples provided by our veterinary partners.
Canine 3D in vitro models – Reconstructed Canine Epidermis (RCE)
For veterinarians, the condition of the dog’s skin gives indications of his general health. In addition, the effect evaluation of dermo-cosmetic products on in vitro reconstructed skin is an essential step for the development of finished products with a topical administration.
QIMA Life Sciences offers Reconstructed Canine Epidermis models that:
- can evaluate the effects of active ingredients and finished products on the epidermis.
- are suitable for evaluations in cases of both systemic or topical administration.
- are used prior to development in order to test a large number of active ingredients and select the best candidates for future clinical studies.
In addition to the “conventional” in vitro models, there are also altered canine models.
QIMA Life Sciences has a long expertise in the development of protocols and the modeling of pathologies and can propose models of induced pathologies (atopic dermatitis, inflammation, infections, senescence, degeneration) or of damaged skin (stress, wound healing).
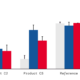
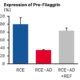
Studies have been conducted on an RCE model of atopic dermatitis induced by a cocktail of cytokines. This model allows the in vitro evaluation of the efficacy of active principles or finished products on dermatological disorders such as AD.
We have carried out morphological / histological analyses of the permeability of the skin barrier, of epidermal proteins and of markers of hydration and inflammation. Click here to view the results of this study.
Find more on our Reconstructed
Canine Epidermis model
ex vivo models of canine whole blood
As part of our whole blood bioassay services, QIMA Life Sciences offers a suite of assays using fresh canine whole blood. Applications of this platform include:
- Bioactivity assays
- Inflammation and immunotoxicity
- Immunogenicity
- Vaccine development: adjuvant, specific immune response
- Allergenicity
For example, we have performed studies to evaluate the immunomodulatory properties of a product on the production of IFNg (activity of T lymphocytes) and on the activation of the COX-2 (PGE2) and 5-LOX (LTB4) pathways, using our ex vivo canine whole blood models. Click here to view some of our results.
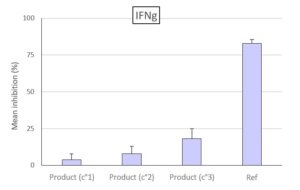
Evaluation of the effect of the tested product (3 concentrations) compared to the reference on the production by T lymphocytes of IFNg
Bioanalysis of non-invasive samples
Our company has developed ready-to-use non-invasive collection kits to analyze surface skin markers (lipids and proteins) from samples from pre-clinical and / or clinical studies.
These bioanalyses make it possible to evaluate:
- The quality of the barrier function of the epidermis (analysis of epidermal lipids and proteins)
- Hydration of the skin (analysis of the components of NMF)
- The skin microbiota
- Markers of inflammation (cytokine quantification)
- Markers of oxidative stress
- Odorous molecules and volatile organic compounds
DID YOU KNOW?
The major assets of in vitro models are their biodiversity and representativeness. While in vivo studies are limited to “standard” animals and to some breeds only, and particularly the Beagle breed for canines, the in vitro approach makes it possible to work on a variety of polymorphisms in terms of age (a particularly important issue for pet food) or of pathological predispositions specific to certain breeds, such as atopic dermatitis and osteoarticular disorders in Labradors.