Wound healing, hemostasis phase and inflammatory phase
Hemostasis phase
The hemostasis phase is the immediate response to injury to prevent blood loss at the wound site in the first few minutes. Damage of capillaries and bleeding triggers a cascade of vasoconstriction and coagulation through extrinsic and intrinsic pathways, leading to platelet aggregation and formation of blood clot consisting of fibrin and fibronectin, which plugs the wound. Platelets also release cytokines, chemokines and growth factors, including PDGF, VEGF and TGF-β, which promote the recruitment and migration of inflammatory cells (neutrophils and macrophages) as well as fibroblasts and endothelial cells to the site of injury.
Infllammatory phase
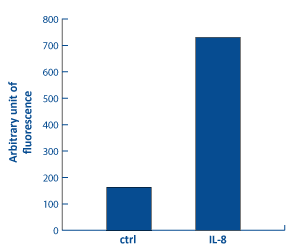
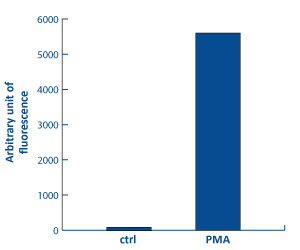
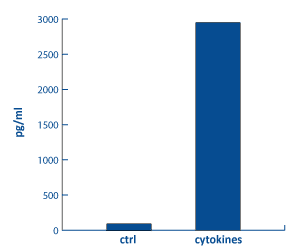
The inflammatory phase is characterized by the sequential infiltration of polymorphonuclear neutrophils (PMNs), monocytes/macrophages and lymphocytes. IL-8 facilitates PMNs migration from surrounding blood vessels [Bioassay: PMN-0009]. By phagocytozing infectious invaders and cellular debris and secreting proteases (matrix metalloproteinases and elastase), PMNs cleanse the wound. They also generate reactive oxygen species [Bioassay: PMN-0005], and together with keratinocytes, release antimicrobial and cytotoxic peptides (AMPs), like β-defensins, RNAse 7, S100A7, and LL-37, thus contributing to host defence [Bioassays: NHEK-00035 ; NHEK-0037 ; EPIBA-0050].
Analysis of PMN migration by
fluorescence based assay
[Bioassay: PMN-0009]
Analysis of H2O2 content in PMN by
fluorescence based assay
[Bioassay: PMN-0005]
Analysis of β-défensin 2 in
epidermal keratinocytes by ELISA
[Bioassay: NHEK-0035]
Meanwhile, monocytes, attracted by chemotactic agents including TGF-β, PDGF, elastin and collagen breakdown products, migrate into the wound and differentiate into mature macrophages. Macrophages continue the cleansing process, fight infections and produce tissue growth factors (FGF, EGF, TGF-β…) as well as inflammatory (TNF-α, IL1-β, IL-8 and IL-6) and anti-inflammatory cytokine (IL-10), which activate keratinocytes, fibroblasts and endothelial cells involved in the next step of repair. More specifically, TGF-βs have a pivotal role in scar formation by acting on cell targets via SMAD and Wnt-dependent pathways, with scarring or anti-scarring effects depending on the TGF-β isoform . Lymphocytes T lastly enter the site >36h after injury.