Com a ajuda dos seus colegas acadêmicos (Laboratoire Inflammation, Tissus Epithéliaux et Cytokines, LITEC, EA 4331) e dos colegas do hospital de Poitiers (University Hospital Center of Poitiers), a QIMA Life Sciences realizou um longo programa de pesquisa sobre psoríase por mais de uma década. Nosso trabalho tem como objetivo especialmente caracterizar melhor a função, além dos efeitos individuais ou combinados de citocinas th1, Th17 e th22 (por exemplo, IL-17A, IL-22, IL-1alfa, OMS e TNF-alfa) no desenvolvimento de inflamação cutânea e da inibição de diferenciação da pele. Hoje a QIMA Life Sciences oferece uma ampla variedade de modelos inovadores e ensaios farmacológicos in vitro especialmente dedicados para a avaliação da eficácia farmacológica (screening, caracterização, prova de conceito) dos seus produtos (APIs, biossimilares, formulações, dispositivos médicos):
- resposta imunológica (liberação de citocina Th17/Th22)
- indução de um perfil semelhante à psoríase nos queratinócitos (ou epiderme reconstruída) por citocinas
- Produção de quimiocinas, peptídeos antimicrobianos (AMPs) e a expressão de marcadores de diferenciação epidérmica
- Análise de marcador específico por imunoensaios ou RT-qPCR
Aqui se encontram alguns exemplos dentre todos os ensaios propostos pela QIMA Life Sciences sobre psoríase:




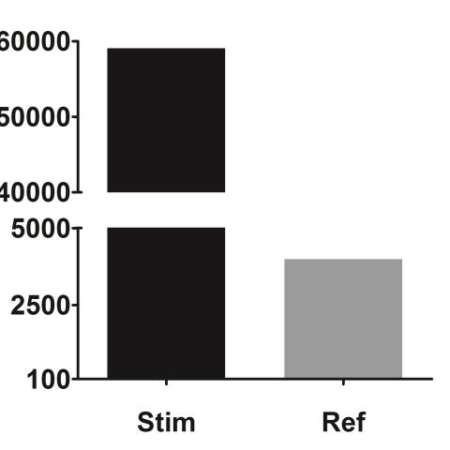
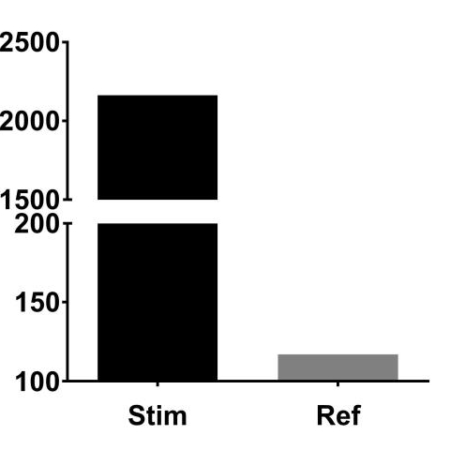
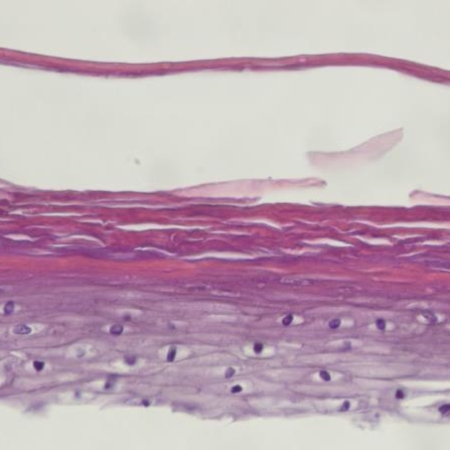
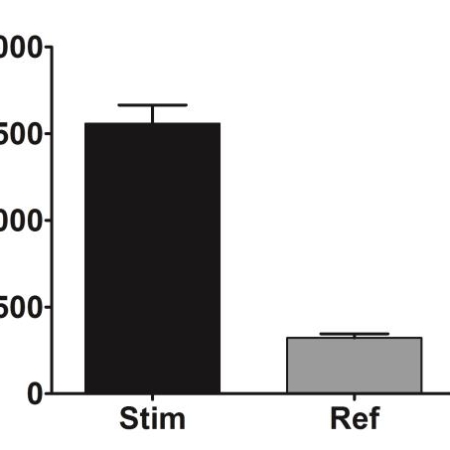
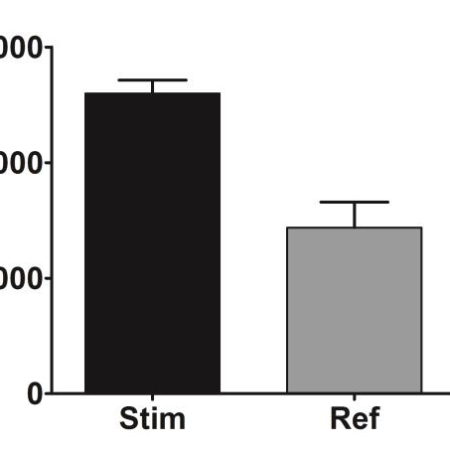


Interleukin-17A-induced production of acute serum amyloid A by keratinocytes contributes to psoriasis pathogenesis
PsoriasisAcute-serum Amyloid A (A-SAA), one of the major acute-phase proteins, is mainly produced in the liver but extra-hepatic synthesis involving the skin has been reported. Its expression is regulated by the transcription factors NF-κB, C/EBPβ, STAT3 activated by proinflammatory cytokines.
Imiquimod-induced skin inflammation in mice is dependent on IL-1R1 and MyD88 signaling but independent of the NLRP3 inflammasome
PsoriasisThe pathogenesis of inflammatory skin diseases such as psoriasis involves the release of numerous proinflammatory cytokines, including members of the IL-1 family. Here we report overexpression of IL-1α, IL-1β, and IL-1 receptor antagonist mRNA, associated to expression of IL-23p19, IL-17A, and IL-22 in skin cells
Inhibition of keratinocyte differentiation by the synergistic effect of IL-17A, IL-22, IL-1α, TNFα and oncostatin M
Inflammation, PsoriasisThis study highlights the precise role of cytokines in the skin inflammatory response (psoriasis). IL-22 and OSM more specifically drive epidermal hyperplasia and differentiation loss while IL-1α, IL-17A and TNFα were more involved in the activation of innate immunity.
Contribution of IL22 to experimental skin inflammation
Inflammation, PsoriasisFocused on in vitro human models, we present the mechanisms of action of IL22 as well as its involvement in structure, metabolism, differentiation, chemotaxis, antibacterial activity, innate immunity, and tissue remodeling of epidermis.
Keratinocytes under fire of proinflammatory cytokines: Bonafide innate immune cells involved in the physiopathology of chronic atopic dermatitis and psoriasis
Atopic dermatitis, Dermatite atopique, PsoriasisSpecific cytokine environment deregulation plays a central role on skin morphology and innate immunity, moving towards specific pathologies and opening the way to new therapeutic strategies.
Skin inflammation induced by the synergistic action of IL17A, IL22, OSM, IL1α and TNFα recapitulates some features of psoriasis
PsoriasisOur results demonstrate the important potentiating activities of IL17A, IL22, oncostatin M, TNF-alpha, and IL1alpha on keratinocytes. This is particularly interesting in the context of psoriasis where these cytokines are overexpressed and could synergize to play an important role in upregulation of chemokines and antimicrobial peptides production.
Cytokine-induced CEACAM1 expression on keratinocytes is characteristic for psoriatic skin and contributes to a prolonged lifespan of neutrophils
PsoriasisThese results show that cytokine-induced cell-surface expression of CEACAM1 by keratinocytes in the context of a psoriatic environment might contribute to the persistence of neutrophils and thus to ongoing inflammation and the decreased propensity for skin infection, typical for patients with psoriasis.
A role for T cell-derived interleukin 22 in psoriatic skin inflammation
PsoriasisThis study indicate that interleukin 22 is a cytokine produced by skin-infiltrating lymphocytes that is potentially involved in initiation and/or maintenance of the pathogenesis of psoriasis.
OSM (Oncostatin M) secreted by skin infiltrating T lymphocytes is a potent keratinocyte activator involved in skin inflammation
PsoriasisThese results demonstrate that OSM and its receptor play an important role in cutaneous inflammatory responses in general and that the specific effects of OSM are associated with distinct inflammatory diseases depending on the cytokine environment.
Keratinocytes as targets for IL10-related cytokines : a putative role in psoriasis pathogenesis
PsoriasisIn this review, we discuss recent knowledge about the effects of cytokines of the IL-10 family on keratinocytes and their potential role in psoriasis, a cutaneous inflammatory disease.
IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes
Inflammation, PsoriasisIL-22 belongs to a family of cytokines structurally related to IL-10, including IL-19, IL-20, IL-24, and IL-26. In contrast to IL-10, IL-22 has proinflammatory activities. IL-22 signals through a class II cytokine receptor composed of an IL-22-binding chain, IL-22RA1, and the IL-10RB subunit, which is shared with the IL-10R.